Sparks, Glencoe, Maryland - April 10, 2026
- ENDOSURE, INC. announces that its ENDOSURE TEST for the rapid and accurate diagnosis of endometriosis and adenomyosis has received CE Marking for commercial use in the European Union. This regulatory milestone enables the company to address the significant unmet medical need for improved endometriosis diagnosis among the approximately 230 million women1 in the European Union.
ENDOSURE, INC.
plans a rapid commercial launch across Europe, supported by its established distribution network to fulfill anticipated orders and deliver the technology to healthcare providers and hospitals throughout the region. The ENDOSURE TEST is already approved for clinical use in the UK, Canada, Brazil, Mexico, the Gulf States, and Israel. The company anticipates a strong market reception and accelerated adoption in Europe.
About Endometriosis
Endometriosis is an incurable, systemic, chronic inflammatory disease. Common symptoms include severe menstrual pain, heavy or abnormal bleeding, chronic pelvic or abdominal pain, infertility, bloating, and nausea. The condition occurs when endometrial-like tissue grows outside the uterus, most commonly in the pelvis, but also potentially in the abdomen, chest, brain, or spine. It affects at least 1 in 4 to 1 in 3 women of reproductive age worldwide2 and can significantly impact quality of life, including sexual intercourse, bowel and bladder function, mental health, and pregnancy outcomes.
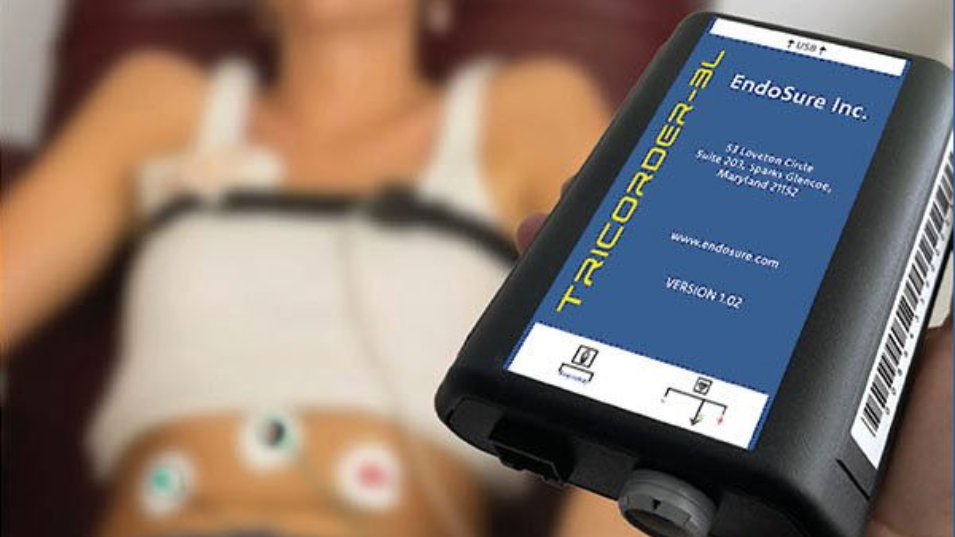
The ENDOSURE TEST is the first and only non-invasive, highly accurate, 30-minute Tier-1 diagnostic test capable of detecting endometriosis at any anatomic location, any stage of disease progression, and at any age, including adolescents and peri- and post-menopausal women. It is also uniquely positioned to document treatment success or failure after surgical or medical therapy and to detect possible disease recurrence. Dr. Mark Noar, company co-founder and inventor of the ENDOSURE TEST, stated: "The importance of this regulatory milestone cannot be measured in words, but rather by the opportunity it creates to eliminate end-stage endometriosis and the associated high costs and destruction of the lives and livelihoods of millions of women, from menarche to post-menopause. For the first time, a non-invasive test has been approved for use in adolescents, where the disease often begins.
"This opens unprecedented opportunities for early suppressive therapy that could conceivably prevent progression to adult end-stage disease within 5 to 10 years." "This significant milestone opens access to one of the world's largest healthcare markets," said Carlos Babini, CEO and co-founder of ENDOSURE INC. "The clinical and economic need for the ENDOSURE TEST has universal appeal. Long diagnostic delays for women are largely driven by limitations in current testing, including cost, access, accuracy, ease of use, and slow results. The ENDOSURE TEST is uniquely positioned to overcome these barriers in Europe through collaboration with leading clinicians, which we believe will drive enthusiastic global adoption.
ENDOSURE, INC. is advancing plans to expand commercial engagement across Europe and to collaborate with leading clinicians, testing centers, and pharmaceutical manufacturers to support evaluation and broader adoption.
About ENDOSURE INC.
ENDOSURE, INC. offers the only non-invasive, 30-minute, highly accurate diagnosis test for endometriosis that not only fills the gap, but also addresses the vacuum in this area of women’s health care.
1 World Population Prospects, United Nations ( UN ), publisher: UN Population Division; Staff estimates, World Bank (WB)
2 de Arruda, Guilherme Tavaresa, et al. Worldwide prevalence of dysmenorrhea: a systematic review and meta-analysis across 70 countries. PAIN 167(1): p 41-55, January 2026