The technology, developed by the 3CPM Company, was one of four finalists to win the National Institute of Health’s RADx Tech ACT ENDO Challenge.
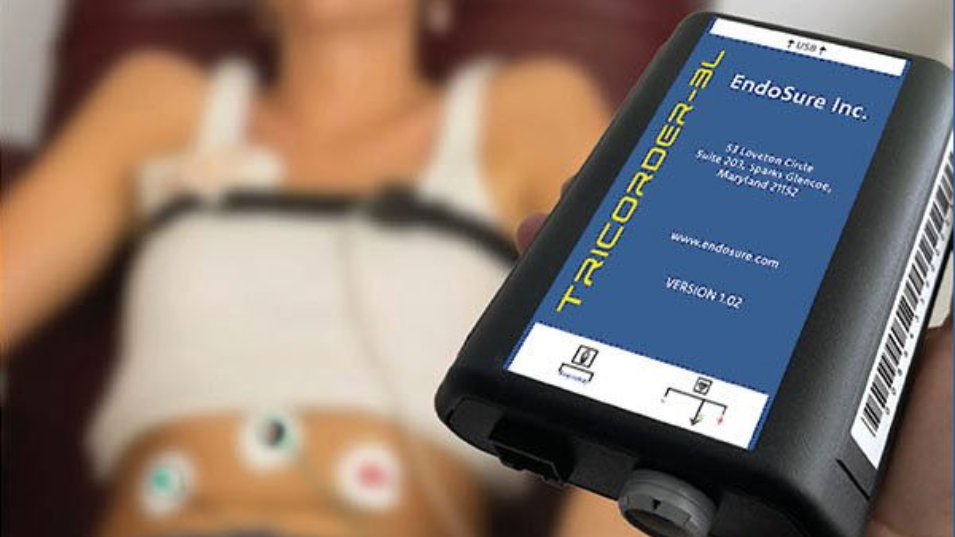
ENDOSURE, INC. is delighted to announce the development of the @HOME version of the award winning ENDOSURE TEST.
The technology, developed by the 3CPM Company, was one of four finalists to win the National Institute of Health’s RADx Tech ACT ENDO Challenge. Once released, the @HOME TEST will make it possible for women to know if they have endometriosis in the comfort of their own homes.
ENDOSURE, INC. holds the exclusive license to globally market this groundbreaking technology.
“3CPM developed, and continues to develop, as well as manufactures the ENDOSURE TEST for us,” said Mr. Carlos Babini, CEO of ENDOSURE, INC. “The positive impact of what our current TEST offers women globally is irrefutable. This new technological advancement promises to be even more life altering. We thank the RADx Tech ACT ENDO Challenge Team along with 3CPM for helping improve lives of women everywhere.”
The ENDOSURE TEST, is the only non-invasive, 30-minute, highly accurate TEST capable of detecting endometriosis in women of any age at any stage, and is currently available in hospitals, clinics, and physicians' offices in select regions of the world (Canada, Brazil, the Middle East, and Europe).
With the development of an at home test, testing will no longer be limited to the clinical environment.
“When we are finished, never again will a 10-year-old with dysmenorrhea have to endure the inconvenience and embarrassment of visiting a gynecologist office to discover if endometriosis is causing her intense painful menstruation,” stated Dr. Mark Noar, inventor of the technology and CEO of 3CPM Company. "Instead, she will use the home ENDOSURE TEST in the comfort of her home with her mother by her side and then share the TEST result with her healthcare provider to determine next steps."
“When we are finished, never again will a 10-year-old with dysmenorrhea have to endure the inconvenience and embarrassment of visiting a gynecologist office to discover if endometriosis is causing her intense painful menstruation." - Dr. Mark Noar, inventor of the technology and CEO of 3CPM Company.
The RADx Tech ACT ENDO Challenge was designed to accelerate the development of innovative, non-invasive diagnostic solutions for endometriosis and reduce the nearly 10-year wait women experience before ever receiving a diagnosis. The Challenge was sponsored by the National Institutes of Health (NIH) Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) and National Institute of Biomedical Imaging and Bioengineering (NIBIB).
About the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD): NICHD leads research and training to understand human development, improve reproductive health, enhance the lives of children and adolescents, and optimize abilities for all. For more information, visit https://www.nichd.nih.gov.
Please join us in celebrating 3CPM Company's significant achievement!


